
Тел.: +7 (383) 330-67-71, факс: +7 (383) 330-80-56, E-mail: bic@catalysis.ru
630090, Россия, Новосибирск, пр-т Ак. Лаврентьева, 5

Тел.: +7 (383) 330-67-71, факс: +7 (383) 330-80-56, E-mail: bic@catalysis.ru
630090, Россия, Новосибирск, пр-т Ак. Лаврентьева, 5
INORGANIC CHEMISTRY: Single crystals of iridium complex can exchange ligands and hydrogenate ethylene.
A SINGLE CRYSTAL of an iridium complex can exchange small-molecule ligands, such as CO and NH3, for N2 and selectively catalyze the conversion of ethylene to ethane, report researchers from the University of North Carolina, Chapel Hill (Nature 2010, 465, 598).
“Reactions of molecular crystals with gases involving covalent bond breaking and formation are as yet quite a rare observation,” says Lee Brammer, a chemistry professor at the University of Sheffield, in England. That the nonporous molecular crystals studied by the UNC group can hydrogenate ethylene is groundbreaking, he adds.
In the new work, Zheng Huang, Peter S. White, and Maurice Brookhart used a bulky, electron-deficient ligand, C6H3[OP(C6H2,(CF3)3)2]2, and N2 to create the square planar iridium complex [Ir]-N2 (shown). Crystallization of the complex from toluene yielded light red single crystals. Structures of the crystals showed that the iridium complex’s four CF3-substituted aryl rings form a pocket around the N2 site. Multiple iridium complexes stack in such a way that disordered toluene molecules line a channel that runs along the smallmolecule site.
When they exposed the [Ir]-N2 crystals to gases of small molecules, Huang and colleagues found that the N2 exchanged to form crystals of different colors: dark red [Ir]-NH3, orange [Ir]-CO (end-on with an Ir-C bond), deep red [Ir]-C2H4 (side-on), green [Ir]-O2 (side-on), and light red [Ir]-(H)2(H2) (two Ir-H bonds and one side-on H2). The crystals’ ability to exchange ligands depends on the size of the incoming molecule. For example, although the crystals took up ethylene to form [Ir]-C2H4, they would not take up propylene.
Huang and coworkers believe that the gases enter the crystal through the channel occupied by the disordered toluenes. Vacuum experiments demonstrated that the small-molecule ligands are not simply lost, leaving an open coordination site for the new ligand to fill. Instead, a ligand-exchange mechanism seems to be at work.
The group also found that when crystals of [Ir]-N2 were exposed to a mix of ethylene, propylene, and hydrogen, ethylene was selectively hydrogenated. That selectivity is not seen in common heterogeneous palladium-on-carbon hydrogenation catalysts, Sheffield’s Brammer notes.
Although growing single crystals would be impractical for large-scale catalysis, details gained from studying this system could provide insight into the catalytic mechanism and lead to better catalyst design, says Paul R. Raithby, a chemistry professor at the University of Bath, in England. Raithby points to measurements of solid-gas reaction kinetics and the possible development of new gas sensors based on single crystals as possible applications of the work. — JYLLIAN KEMSLEY
WWW.CEN-ONLINE.ORG
JUNE 7, 2010
ChemCatChem
Mechanistic Studies on Hydrocyanation Reactions
Laura Bini, Christian Müller, Dieter Vogt
Magnetic Nanocomposites: A New Perspective in Catalysis
Yinghuai Zhu, Ludger Paul Stubbs, Feny Ho, Rongzhen Liu, Chee Peng Ship, John A. Maguire, Narayan S. Hosmane
Highly Enantioselective Resolution of β-Amino Esters by Candida antarctica Lipase A Immobilized in Mesocellular Foam: Application to Dynamic Kinetic Resolution
Mozaffar Shakeri, Karin Engström, Anders G. Sandström, Jan-E. Bäckvall
The Catalytic Conversion of Thiophenes over Large H-ZSM-5 Crystals: An X-Ray, UV/Vis, and Fluorescence Microspectroscopic Study
Marianne H. F. Kox, Ana Mijovilovich, Jesper J. H. B. Sättler, Eli Stavitski, Bert M. Weckhuysen
ChemSusChem
Transition Metal–Tungsten Bimetallic Catalysts for the Conversion of Cellulose into Ethylene Glycol
Ming-Yuan Zheng, Ai-Qin Wang, Na Ji, Ji-Feng Pang, Xiao-Dong Wang,Tao Zhang
Embedded Phases: A Way to Active and Stable Catalysts
Loredana De Rogatis, Matteo Cargnello, Valentina Gombac, Barbara Lorenzut, Tiziano Montini, Paolo Fornasiero
Water-Tolerant Mesoporous-Carbon-Supported Ruthenium Catalysts for the Hydrolysis of Cellulose to Glucose
Hirokazu Kobayashi, Tasuku Komanoya , Kenji Hara, Atsushi Fukuoka
Methyltrioxorhenium Catalysis in Nonconventional Solvents: A Great Catalyst in a Safe Reaction Medium
Marcello Crucianelli, Raffaele Saladino, Francesco De Angelis
Advanced Synthesis & Catalysis
Efficient Heterogeneous Dual Catalyst Systems for Alkane Metathesis
Zheng Huang, Eleanor Rolfe, Emily C. Carson, Maurice Brookhart, Alan S. Goldman, Sahar H. El-Khalafy, Amy H. Roy MacArthur
Regioselective Reactions on a Chiral Substrate Controlled by the
Configuration of a Chiral Catalyst
Raju Ranjith Kumar, Henri B. Kagan
Asymmetric Synthesis with Silicon-Based Bulky Amino Organocatalysts
Li-Wen Xu, Li Li, Zhi-Hui Shi
Efficient Tandem Biocatalytic Process for the Kinetic Resolution of
Aromatic -Amino Acids
Bian Wu, Wiktor Szyma ski, Stefaan de Wildeman, Gerrit J. Poelarends, Ben L. Feringa, Dick B. Janssen
Angewandte Chemie IE
Highly Active Mesoporous Nb-W Oxide Solid-Acid Catalyst
Caio Tagusagawa, Atsushi Takagaki, Ai Iguchi, Kazuhiro Takanabe, Junko N. Kondo, Kohki Ebitani, Shigenobu Hayashi, Takashi Tatsumi, Kazunari Domen
Iron-Catalyzed Direct Arylation of Unactivated Arenes with Aryl Halides
Wei Liu, Hao Cao, Aiwen Lei
Palladium-Catalyzed Aerobic Oxidative Carbonylation of Arylboronate Esters under Mild Conditions
Qiang Liu, Gang Li, Jun He, Jing Liu, Peng Li, Aiwen Lei
Chemistry – A European Journal
Unifying Metal and Bronsted Acid Catalysis—Concepts, Mechanisms, and Classifications
Magnus Rueping, Rene M. Koenigs, Iuliana Atodiresei
Asymmetric One-Pot Sequential Organo- and Gold Catalysis for the
Enantioselective Synthesis of Dihydropyrrole Derivatives
David Monge, Kim L. Jensen, Patrick T. Franke, Lennart Lykke, Karl Anker Jorgensen
Nanoparticles as Semi-Heterogeneous Catalyst Supports
Alexander Schätz, Oliver Reiser, Wendelin J. Stark
Chemistry – An Asian Journal
Bromine-Catalyzed Aerobic Oxidation of Alcohols
Muhammet Uyanik, Ryota Fukatsu, Kazuaki Ishihara
Recent Advances in Multicatalyst Promoted Asymmetric Tandem
Reactions
Jian Zhou
Helvetica Chimica Acta
Copper-Catalyzed Ritter-Type Reaction of Unactivated Alkenes with
Dichloramine-T
Takumi Abe, Hiroyuki Takeda, Yoshihisa Miwa, Koji Yamada, Reiko Yanada, Minoru Ishikura
Pronounced Catalytic Activity of Manganese(III)–Schiff Base Complexes in the Oxidation of Alcohols by Tetrabutylammonium
Peroxomonosulfate
Abdolreza Rezaeifard, Maasoumeh Jafarpour, Mohammad Ali Nasseri, Reza Haddad
Chinese Journal of Chemistry
Water Soluble Starch Stabilized Palladium Nanoparticle: Efficient
Catalyst for Miyaura-Suzuki Coupling Reaction
Shiyong Liu, Qizhong Zhou, Huajiang Jiang
Fast and Eco-friendly Synthesis of Dipyrromethanes by H2SO4•SiO2
Catalysis under Solvent-free Conditions
Yan Zhang, Jun Liang, Zhicai Shang
ZAAC – Zeitschrift für anorganische und allgemeine Chemie
The Coupling of Pyridine and Dichloromethane Mediated by UO2Cl2
Robert J. Baker, Emtithal Hashem, Majid Motevalli, Helen V. Ogilvie, Aurora Walshe
Quinine-Derived Imidazolidin-2-imine Ligands: Synthesis, Coordination Chemistry, and Application in Catalytic Transfer Hydrogenation
Sabina-Alexandra Filimon, Cristian G. Hrib, Sören Randoll, Ion Neda, Peter G. Jones, Matthias Tamm
European Journal of Organic Chemistry
When Organocatalysis Meets Transition-Metal Catalysis
Volume 2010, Issue 16, June 2010, Pages: 2999–3025
Chemical Engineering & Technology
In situ Monitoring of Coke Deposits during Coking and Regeneration of Solid Catalysts by Electrical Impedance-based Sensors
Norbert Müller, Ralf Moos, Andreas Jess
Chemie Ingenieur Technik
Zeolithkatalyse für die nachhaltige Chemie
Wladimir Reschetilowski
Berichte zur Wissenschaftsgeschichte
(Re)cognizing Postmodernity: Helps for Historians - of Science Especially
Paul Forman
Impact Factors
Category: ENGINEERING, CHEMICAL, Sorted by Impact Factor
| Rank | Title | 2009 Impact Factor |
| 1 | PROGRESS IN ENERGY AND COMBUSTION SCIENCE | 11.024 |
| 2 | JOURNAL OF CATALYSIS | 5.288 |
| 3 | APPLIED CATALYSIS B-ENVIRONMENTAL | 5.252 |
| 4 | CATALYSIS TODAY | 3.526 |
| 5 | PROCEEDINGS OF THE COMBUSTION INSTITUTE | 3.256 |
| 6 | CHEMISTRY AND PHYSICS OF CARBON | 3.250 |
| 7 | JOURNAL OF MEMBRANE SCIENCE | 3.203 |
| 8 | FUEL | 3.179 |
| 9 | COMBUSTION AND FLAME | 2.923 |
| 10 | SEPARATION AND PURIFICATION TECHNOLOGY | 2.879 |
| 11 | DYES AND PIGMENTS | 2.855 |
| 12 | CHEMICAL ENGINEERING JOURNAL | 2.816 |
| 13 | AEROSOL SCIENCE AND TECHNOLOGY | 2.739 |
| 14 | JOURNAL OF SUPERCRITICAL FLUIDS | 2.639 |
| 15 | SEPARATION AND PURIFICATION REVIEWS | 2.615 |
| 16 | JOURNAL OF AEROSOL SCIENCE | 2.529 |
| 17 | REACTIVE & FUNCTIONAL POLYMERS | 2.461 |
| 18 | PROCESS BIOCHEMISTRY | 2.444 |
| 19 | FUEL PROCESSING TECHNOLOGY | 2.321 |
| 20 | ENERGY & FUELS | 2.319 |
Category: ENGINEERING, CHEMICAL, Sorted by Citations
| Rank | Title | 2009 Total Citations |
| 1 | JOURNAL OF CATALYSIS | 31,744 |
| 2 | INDUSTRIAL & ENGINEERING CHEMISTRY RESEARCH | 25,480 |
| 3 | JOURNAL OF MEMBRANE SCIENCE | 24,096 |
| 4 | CHEMICAL ENGINEERING SCIENCE | 21,512 |
| 5 | CATALYSIS TODAY | 20,194 |
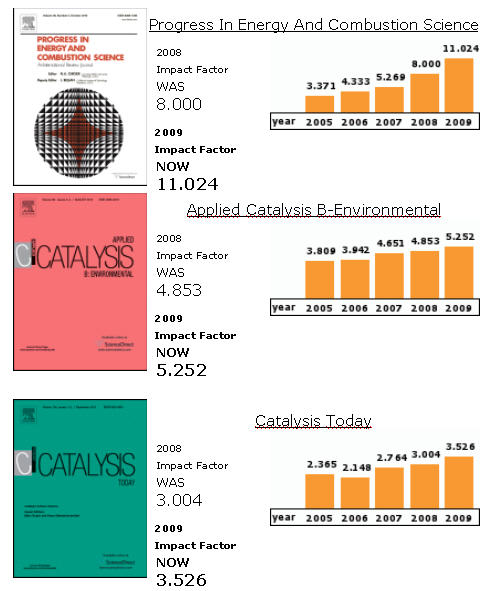

Journal of Catalysis, Applied Catalysis B: Environmental and Catalysis Today are ranked 2nd , 3rd and 4th respectively within Thomson Reuters' Engineering, Chemical category.*
Journal of Catalysis is also ranked 1st in the Top Most Cited Journals in Engineering, Chemical.
| Title | 2009 Impact Factor* |
| Applied Catalysis A, General | 3.564 |
| Applied Catalysis B: Environmental | 5.252 |
| Catalysis Communications | 3.000 |
| Catalysis Today | 3.526 |
| Chinese Journal of Catalysis | 0.786 |
| Journal of Catalysis | 5.288 |
| Journal of Hazardous Materials | 4.144 |
| Journal of Molecular Catalysis A: Chemical | 3.135 |
| Journal of Molecular Catalysis B: Enzymatic | 2.400 |
| Microporous and Mesoporous Materials | 2.652 |